Thanks for the reply. And I’m not meaning to imply you did not do your homework, sorry if it comes accross that way.
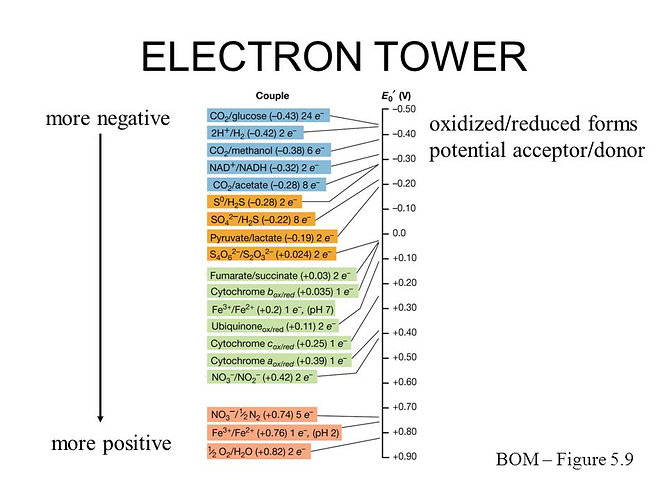
Oh yes, I know oxidising does not require oxygen, a large part of my education has also been in microbiology. I was just aluding to the origin of the word. Regardless, without oxygen you need another compound to “catch” the electrons for you. In order for a cell to derive energy from a reaction, there must be a potential difference in the right direction between the electron source (f.e. Fe2+) and the electron sink (f.e. O2). This is commonly visualized in the electron tower:
Basically in order to get energy from moving one compound to the left (oxidising it) you need to move another reaction lower on the tower to the right (reducing it). From what I remembered only really oxygen can really be used to oxidize Fe2+ but your own source offered that other possibility, I’ll have to look into it. But CO2 can definitely NOT be used to oxidize the iron. The autotroph part of chemoautotroph just means they produce their own organic compounds for growth from CO2, like plants do. (So CO2 is part of the growth process, not part of the producing energy process)
From the article you linked, you should keep in mind that chemolithotrophs are a broad group, and just because some of them do not use O2, does not mean the iron-oxidising ones don’t need it. As you can see in the tower, getting energy from iron is a lot harder than the others.