Here I am once again repeating my vision and gathering information about topics that are not talked about much.
This time I want to talk about several things that I notice that are largely ignored (except for one that I will talk about here), the points that should be addressed, and ideas that occurred to me while writing, or before.
First of all, a few notes:
- Of course my English is not the best and I use a third party (this time with the help of Grammarly)
- I collected information on quite similar topics that were discussed in the past:
Links
Compounds and Compound Toxicity - #5 by Deus
A Stress System, DNA Point Gaining and Temperature Control
Homeostasis and Other Metabolic Pathways
Acid organelles
Ideas for Part Ugrades and Mechanics - #8 by DissonanceFall
- I am here presenting scientifically as far as possible why this is so and it is the responsibility of the readers to think for themselves if it is true for the game that there was something like this.
The battles of early life - Introduction and concepts
Since the beginning of the narrowness of life, which no one yet knows, they had several enemies that threatened to end what had not yet begun, well-known examples such as radiation, lack of organic materials and available energy, and even water itself were their enemy.[1]
But today I am not going to talk about the beginning of life or what is related to it, those are for another battle since one thing is linked but it is not charged enough and there is a missed opportunity: osmosis. Or more correctly in this case - the concentration of ions in the environment.
Vs salts
First of all, for those who are familiar with the seawater that is always salty or the table salt that we add to our food, this is one of the types of salt that I am talking about: sodium, chlorine, potassium, and calcium ions are the most familiar examples of free ions in the sea.
Where did they come from? Some came from water eroding rocks and transporting them to the oceans, but there is a study that says this is not a sufficient amount to explain why there is a lot of salt in the sea in general and its origin comes from volcanic activity[2] that with each eruption brought to the oceans salts and important substances, which in the future began to finance the life that began to live in it.
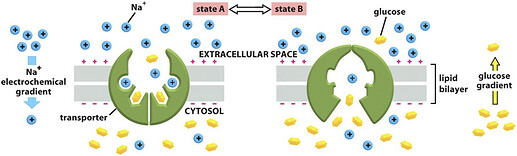
And these salts play a very important role in many processes that take place in the cells: such as the use of sodium ions in the process of introducing glucose into the cells:
and participation in processes inside the cells that take too long for me to detail here.[3]
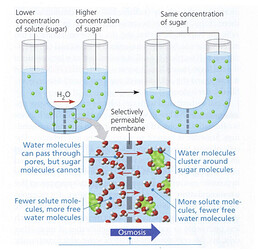
But there is also something else and this is where the concept that exists in the game “osmoregulation” comes into play: the first war of the cells against chemistry and physics - concentration cascade, or osmosis.
According to a law that exists in nature - diffusion, a passive process does not “consume” energy when materials move to a place where their concentration is lower. This is how your coffee mixes. But there is also a second case, and this is a very important process in life and enters into our game - osmosis:
When there is a selective membrane, water will move to the place where the concentration is greater and balance the concentrations. This process can be beneficial or harmful to the cell.
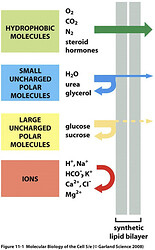
And the reason for this is that they are ionic:
Ions are unable to cross the membrane and rely on channels and carriers whose job is to move them into the cell. I’m not going to go into detail about it here because it would take too much time and it’s unnecessary, but the reason I bring it up is that you need this knowledge to understand what I’m going to explain here.
Vs Temperature
Life has always fought against temperature changes - from the volcanic heat of the underwater emanations to the ice shelves of the poles and the change in temperature from the atmosphere and space, these are challenges that even today we feel if climate change and it greatly affects life.
According to what we know from science, at the beginning of life, the surface temperature of the Earth was around 85 degrees Celsius (or 358 Kelvin). [4]
If this is true, it means that we are the extreme ones in terms of our adaptation to a lower temperature than the first life was and not the archaeans found in volcanic lakes in Yellowstone. But on the other hand, life must adapt to different temperatures, because proteins and various organic substances behave differently at each temperature. If the temperature is too low, they will activate too slowly for them to function at a good rate, and if it is too hot, the proteins will be denatured as in hard-boiled eggs.
This is also the disadvantage of homothermic (“warm blood”) compared to pelcolothermics (“cold blood”) whose proteins are adapted only to this specific temperature.
But life can find a way to deal with it and there are many tricks up its sleeves, one of which I will explain later.
Vs PH
Acids and bases are something that nature and life itself have always revolved around.
On the one hand, they help us digest and disinfect food, and maintain the proper functioning of important activities and feedback in the body (blood with carbon dioxide or the thickness of the skin, etc.). But on the other hand, acids and bases are very active radicals that can severely damage organic molecules and change them, making them useless or extremely dangerous.
In ancient life, their concentration was very wide and this is a serious problem for the cells that lived at that time.
[5]
The cells have developed many mechanisms that help to balance or protect against such changes. But today, these conditions are rare and the pH in the sea is usually around 7 - 8, but this changes and becomes acidic due to the increase in carbon dioxide concentrations. It already has a big impact on life there. Some of the issues I’m going to focus on were related to this.
And now that we’ve summarized their concepts and effects (briefly), I’m now going to explain my ideas for the game
Walls and membranes facing a changing world
First of all, I want to talk about one thing that bothers me a little about the whole thing the realism of the game (I’m not against anyone, and this is my opinion):
Calcium carbonate and silica.
Leaving their pros and cons aside, I’m going to talk about one more important thing if you’ve been paying attention to the entire introduction - they’re not organic materials.
Calcium carbonate is a carbonaceous molecule with calcium ions that have been synthesized and given a significant advantage when it comes to protection and stability. Many living things use some form of calcium and this is where I get to my point - to make them, the creature needs to absorb calcium ions to make them.[6]
First of all, I want to talk about one thing that bothers me a little about the whole thing about the realism of the game (I’m not against anyone, and this is my opinion):
Calcium carbonate and silica.
Leaving their pros and cons aside, I’m going to talk about one more important thing if you’ve been paying attention to the entire introduction - they’re not organic materials.
Calcium carbonate is a carbonaceous molecule with calcium ions that have been synthesized and given a significant advantage when it comes to protection and stability. Many living creatures use some form of calcium (mollusks, crustaceans, even vertebrates or foraminifera) and here I get to my point - to create them, the creature needs to absorb calcium ions to create them (in marine creatures).
Even this is one of the first defenses that multicellular life invented when carnivores appeared. But there is one small problem - any change in the environment affects these creatures especially drastically.
One significant factor is acid, and there is evidence that due to the disappearance of creatures with calcium carbonate, which make up the basis of our ecology on Earth, contributed to the worsening of extinctions in the oceans[7] [8]
Although I’m talking here about something that seems somewhat unrelated to what I’m talking about: walls and membranes, I see here an opportunity to do something a little unique - my idea is simply in certain cases such as events, to lock them and prevent them from using them because the conditions are not suitable, or Warn them that playing with them will make it difficult for them. This will add a challenge and also a milestone in their choice - whether to change the membrane to something else temporarily or to challenge themselves in this choice. I see the use of these membranes as not so much fun for the game (as someone who likes speeds a bit) and gives them some advantages in certain points - lower cost of buying them when the calcium concentration is high and vice versa.
I think it could also work on its twin - Silica, which also falls under these definitions.
Membrane stability against temp
Another thing I notice, but I guess they are working on it, is temp.
Leave aside the effects and planning changes. Here I want to add one thing realistic about one thing - the fluidity and stiffness of the membrane. In my academic studies, I learned that the structure of the membrane is indirectly related to changes in the temperature of the environment.
For example - in a cold environment, one of the ways to deal with freezing is that the phospholipids that make up the membrane are replaced by unsaturated fats (in the sense of phospholipids), contributing to it becoming more liquid and thus preventing it from freezing. [9]
So I suggest something like this - to add to the option of changing the stability of the membrane so that it affects the resistance of the cell against the temperature (in addition to improving, not as a substitute). Can lead to new strategies for the player and open up new possibilities and make the game a little more interesting than a game of life and speed.
New early energy source - Osmotrophy
Here I want to offer something new that has not been discussed before - since at least my last article on the kinetosynthesis that you invited to see here:
But here its energy source comes from a source that is theoretically very possible in places where the concentration cascade is very strong - a meeting of underwater currents (such as the meeting between the Gulf Stream and the Arctic Current) or in the currents of volcanic emanations, where salts mix and create changes in concentrations. In the invention of using the physical properties explained earlier, this property can be used as a source of energy. It is also an idea that has been presented as a source of energy for possible life forms on Jupiter’s moon Europa. [10]
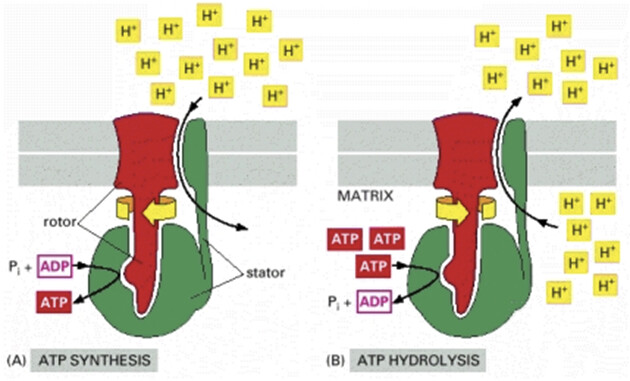
In the findings, perhaps a special protein called (a name I invented) “Osmosome” that in areas where there is a high concentration of ionic cascades can generate energy in the form of ATP, similar to a thermoplastic.
This idea is not far from reality, as there is something similar in our cells called an F-type proton pump:
Only instead of ions, a flow of protons as a source of energy. I’m just expanding on that idea.
Events around pressure and acids
I’ve touched on them in previous topics, but I want to expand a bit more and talk about something else that exists but I don’t see it affecting the game at all in its current state (as of this writing): Atmospheric pressure.
According to several articles, their effect on cells is quite minimal up to 1000 atm (one thousand atmospheres) [11]
And I guess it was more significant the bigger the creature (both at the level of cells and the level that it starts to become multicellular). So I guess pressure can be used as another form of use for it - pressure can be used as an event state in the game.
For example, the pressure caused by the water on the earth can cause the development of new volcanic areas or vice versa. Pressure can perhaps also shape how sand is compressed and turned into stone, creating new areas such as underwater stone deserts and more…
But here I was talking about pressure as literal and not as a game mechanic because I don’t see them at the moment because it doesn’t affect that much now either. The only humidity that affects the cellular level is osmotic pressure (high, low, or unstable concentration) and it is probably the pressure that should be focused on.
Alternatively, maybe adding new areas where the pressure is much higher, for example, underwater canyons (like Marina Challenger) can give it the push it needs.
Another additional point I talked about is acid and the environment. But I want to add something else about it:
Acids as an event can be used as a horizon expander if used correctly. In the P-T extinction event (or the great death), due to an increase in volcanic materials such as volcanic ash and sulfur, caused a decrease in the level of acidity in the water, killing more than 90% of all marine creatures that lived in it.[12]
What I am trying to convey here is that PH in water should be considered an important focus for some of the events that will be added to the future.
Summary
- A short explanation about osmosis, salts, and ions, and acids in ancient life
- To give additional meaning to the calcium carbonate and silica type membrane
- Membrane intervention on temperature resistance
- A new type of energy source is possible according to what was discussed
- The participation of pressure and acid in future events
Conclusions
I guess that was my opinion on a few places that I haven’t seen talked about, and they are equally important on how oxygen and light affect. In addition, I have given some ideas from me about these topics. I suppose it is also possible that they can be discussed here.
And I hope it contributed to the game in some form or another because I feel these married people have been undersaid even though they play an essential role in life.
I also wrote this because I finished the exam on these topics and this is a good opportunity to talk about them as long as the material on the topic of cell biology is still fresh with me XD
https://www.futurelearn.com/info/courses/exploring-our-ocean/0/steps/730 ↩︎
Constraining the climate and ocean pH of the early Earth with a geological carbon cycle model | PNAS ↩︎
(PDF) Effects of Cd2+ on the calcium metabolism and shell mineralization of bivalve Anodonta cygnea (researchgate.net) ↩︎
Ocean Acidification Leaves Mollusks Naked and Confused | Inter Press Service (ipsnews.net) ↩︎
(PDF) Impacts of ocean acidification on marine shelled mollusks (researchgate.net) ↩︎
Cell Membrane Fluidity: Factors That Influence and Increase the Cell Membrane Fluidity – Moosmosis ↩︎
https://books.google.it/books?id=b7XhonTq2s8C&pg=PA185&lpg=PA185&dq=Thermotroph&source=bl&ots=G4-YqVGjUl&sig=2oMSs-lUKEx9XgFOZIkvYFR-I4M&hl=it&sa=X&ei=cGqCUYXGDMvP4QS5zIAw#v=onepage&q="Osmotic%20and%20ionic%20gradients"&f=false ↩︎
(PDF) Effects of high hydrostatic pressures on living cells: A consequence of the properties of macromolecules and macromolecule-associated water (researchgate.net) ↩︎
Ocean acidification killed off more than 90 percent of marine life 252 million years ago, scientists believe | The Independent | The Independent ↩︎